Vietnam approves Russian Sputnik V COVID-19 vaccine
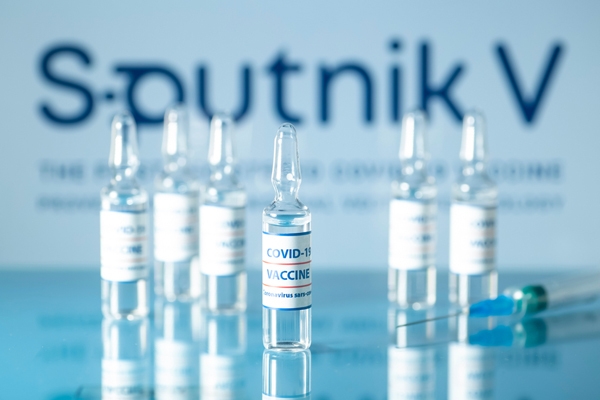 |
| As a preliminary result, two doses of Sputnik V were found to be highly effective and responded well to volunteers over 18 years of age |
Sputnik V COVID-19 vaccine, also named Gam-COVID-Vac, has been conditionally approved due to the urgent need in the prevention and control of the COVID-19 pandemic. This is the second COVID-19 vaccine approved by Vietnam, after the AstraZeneca vaccine of the United Kingdom.
According to the results of the interim analysis of phase 3 trials published in The Lancet, the vaccine was 91.6 per cent effective. As a preliminary result, two doses of Sputnik V are highly effective and responded well to volunteers over 18 years old. It is one of the three COVID-19 vaccines in the world that are more than 90 per cent effective (AstraZeneca, Pfizer-BioNTech, and Moderna COVID-19 vaccine).
On March 16, a batch of 1,000 Sputnik V vaccines was presented to Vietnam by Russia on the occasion of the working trip of the Secretary of the Security Council of the Russian Federation, Nikolai Patrushev.
Shortly after arriving at Noi Bai airport, the patch of 1,000 doses of Sputnik V vaccine were transferred to cold storage at the National Institute of Hygiene and Epidemiology by the Ministry of Health.
Sputnik V has been approved for use in more than 50 countries. Last December, Belarus became the first country outside Russia to approve Sputnik V. In Europe, where the vaccine campaign was slow due to lack of supply, officials began to look to Sputnik V. On March 4, the European Medicines Agency (EMA) began a performance evaluation for the product. In Southeast Asia, the vaccine has been approved in Laos and Myanmar.
This is the second COVID-19 vaccine approved urgently by Vietnam, after the vaccine by AstraZeneca. So far, 117,600 doses of AstraZeneca vaccine were delivered to Vietnam in mid-February to be used from March 8 to vaccinate priority groups. To date, more than 36,000 people have been vaccinated with the AstraZeneca vaccine.
What the stars mean:
★ Poor ★ ★ Promising ★★★ Good ★★★★ Very good ★★★★★ Exceptional
 Tag:
Tag:
Themes: Healthcare Platform
Related Contents
Latest News
More News
- Ministry of Health issues list of free regular health check-ups (May 13, 2026 | 18:34)
- Health-related community campaign launched in Hanoi (May 13, 2026 | 18:29)
- Health Ministry to implement free health check-ups (May 08, 2026 | 09:00)
- Revised Capital Law officially passed (April 24, 2026 | 10:20)
- Hanoi mulls time-based petrol motorbike ban on 11 streets (April 23, 2026 | 17:16)
- Government launches regular health checks alongside food safety crackdown (April 22, 2026 | 10:18)
- Vietnam recognised among top performers in World Bank human capital index (April 20, 2026 | 20:32)
- VCCI refutes US concerns over overcapacity and forced labour in Vietnam (April 17, 2026 | 17:22)
- Vietnamese among Asia’s most optimistic spenders (April 14, 2026 | 16:49)
- MoH urges hospitals to accelerate electronic medical records (April 13, 2026 | 14:19)






















 Mobile Version
Mobile Version